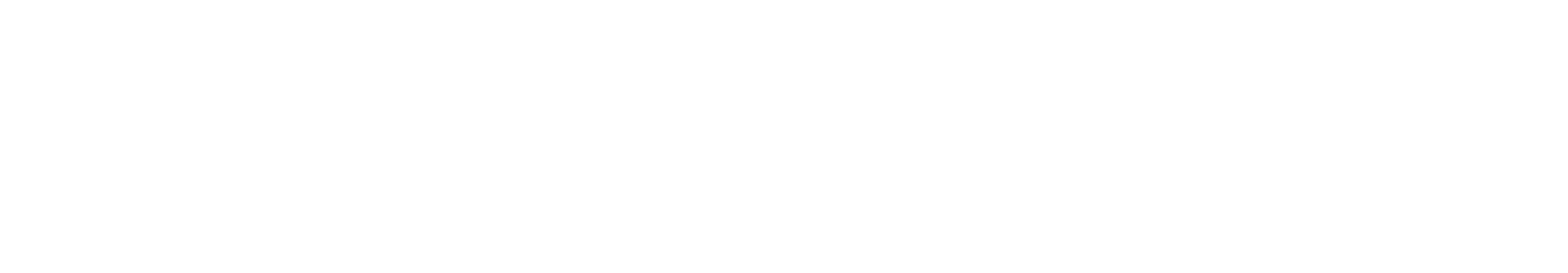Our Services
Quality Management Systems
A QMS must support, not hinder, your development project. We don’t just build scalable systems tailored to company stage, product classification, and risk level. We educate our clients, so they know exactly when is the right time and what are the right procedures to implement to ensure compliance and avoid overspending and overburdening their team.

Capabilities:
- QMS implementation strategies for early development
- Full ISO 13485, 21 CFR 820/QMSR, MDSAP-compliant systems
- SOP development, gap assessments, training
- Risk Management and design controls (IEC 62304, ISO 14971 principles)
- Internal audits and external audit support
Quality Management – Roll out approach